slp.blue®
Modern document and quality management system
slp.blue® is a modern web application for document and quality management. It facilitates the management of documentation with a focus on the specific needs of hospitals and laboratories, including support for creating interlinked documents, dynamic generation of laboratory manuals, and the implementation of other quality management processes. It streamlines documentation workflows for quality managers, document administrators, and other staff. It also helps laboratories comply with the requirements for quality management systems according to ISO 15189:2022.
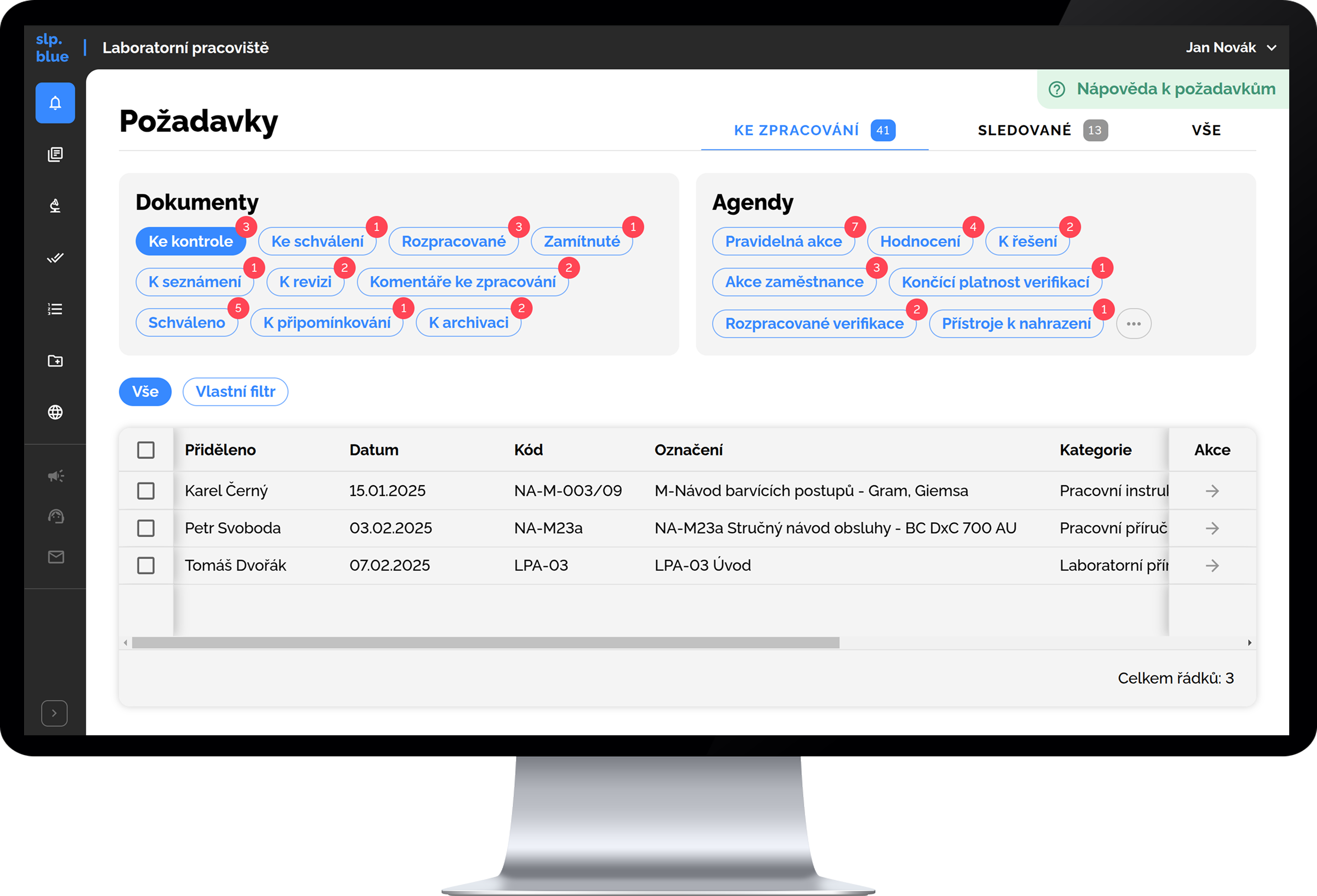
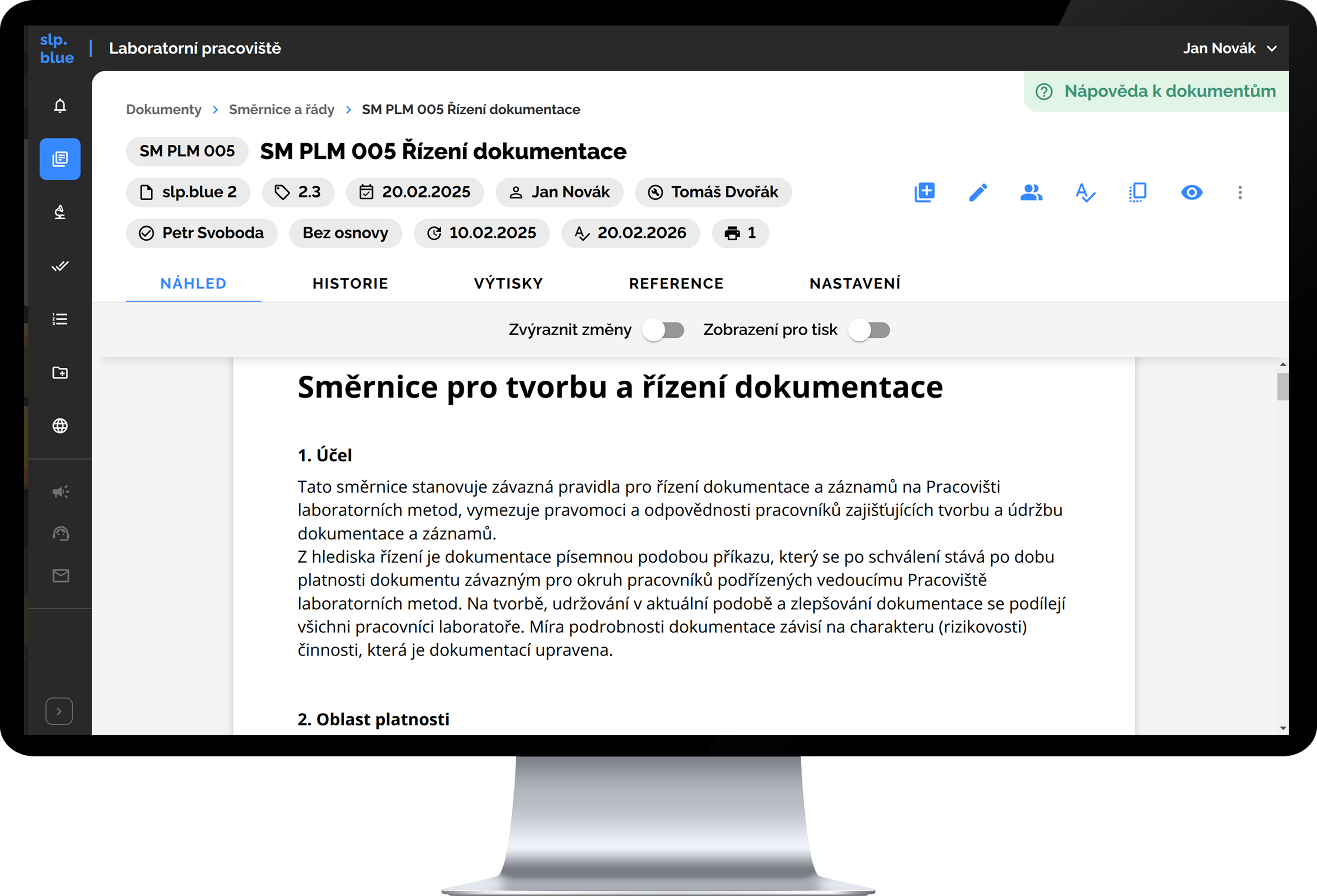
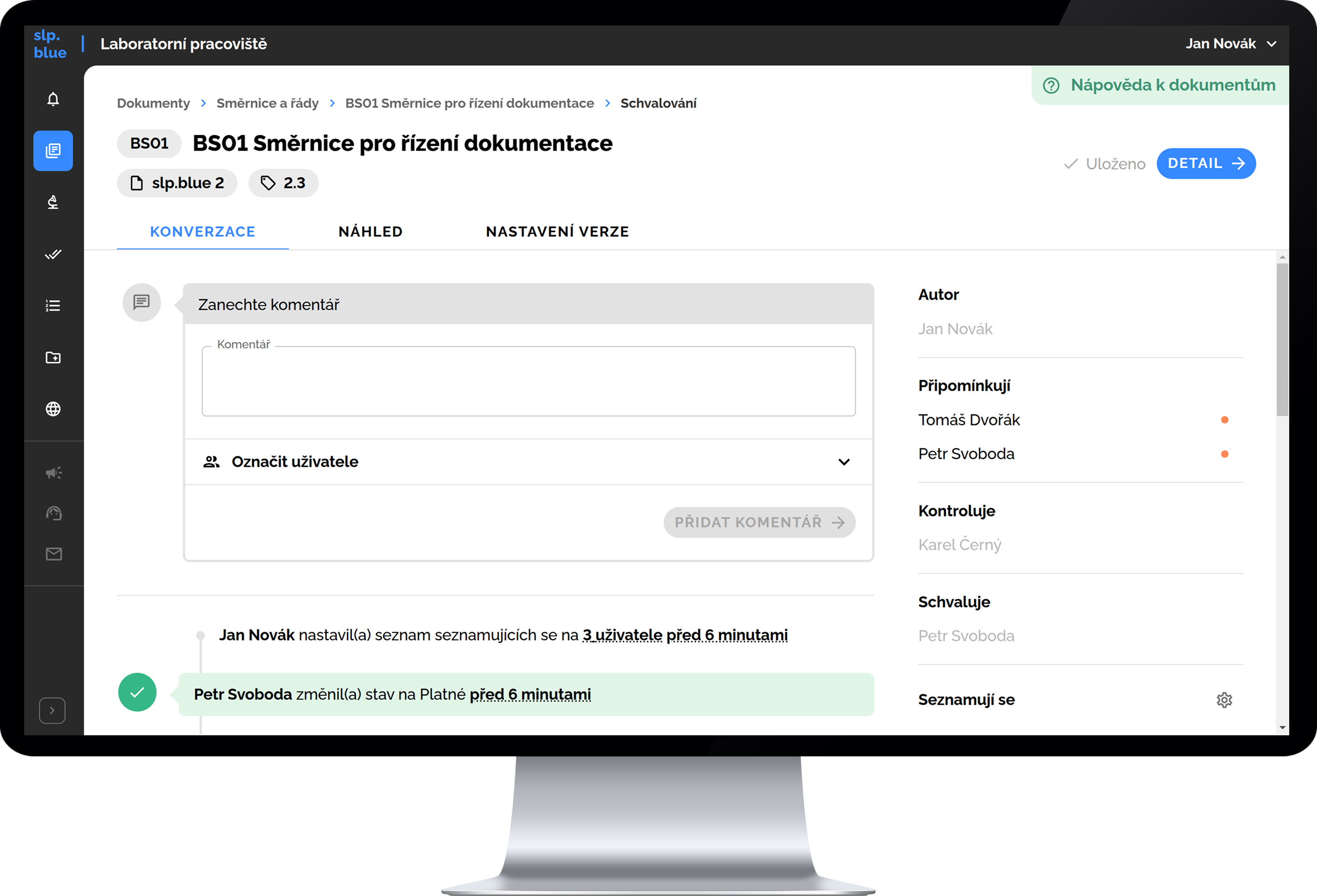
slp.blue includes complete functionality for electronic document management, including version control for individual documents, management of the review and approval process for new versions, setting validity dates, and managing the process of familiarizing users with document changes. It also supports printing documents, including formal elements such as a cover page and header, as well as subsequent management of individual printed copies (e.g., copy number, quantity, location, version, and owner).
Why choose slp.blue
Seamless Compliance with ISO 15189:2022 and IVDR
- Fully aligned with ISO 15189:2022
- Whether you are new to ISO or upgrading from ISO 15189:2012 — we make the transition simple
- Ready to support your IVDR implementation
- Printing optional — fully compliant without paper
Smart Electronic Document Management
- no formatting issues nor compatibility problems
- documents stay interconnected, structured and always up to date
- user acknowledgment of document changes built-in
- full-text search across documents — not just titles or tags.
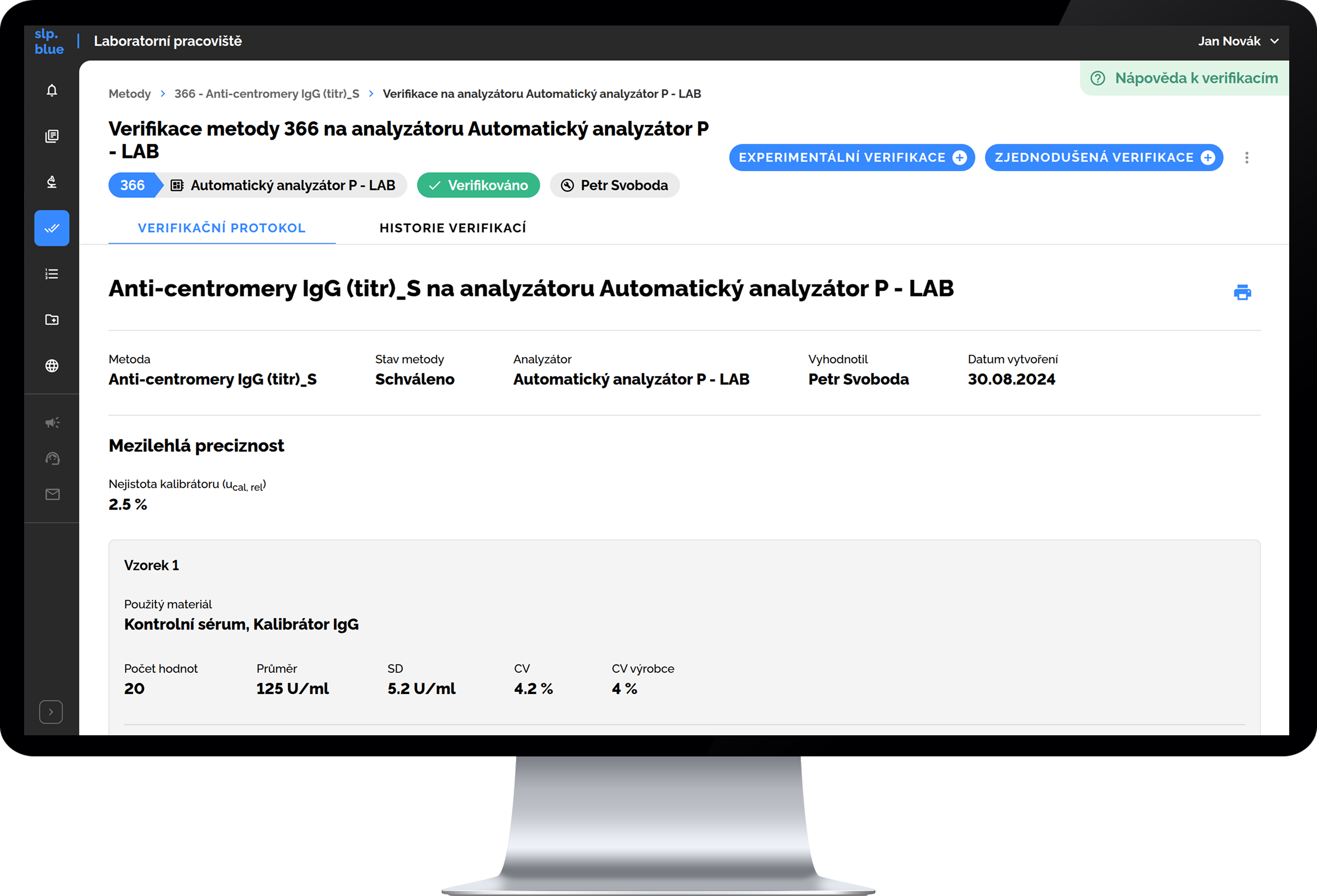
Method Management, Verification & Quality Control
Supports integration with local, national, and international laboratory coding standards — automatically updated and fully structured.
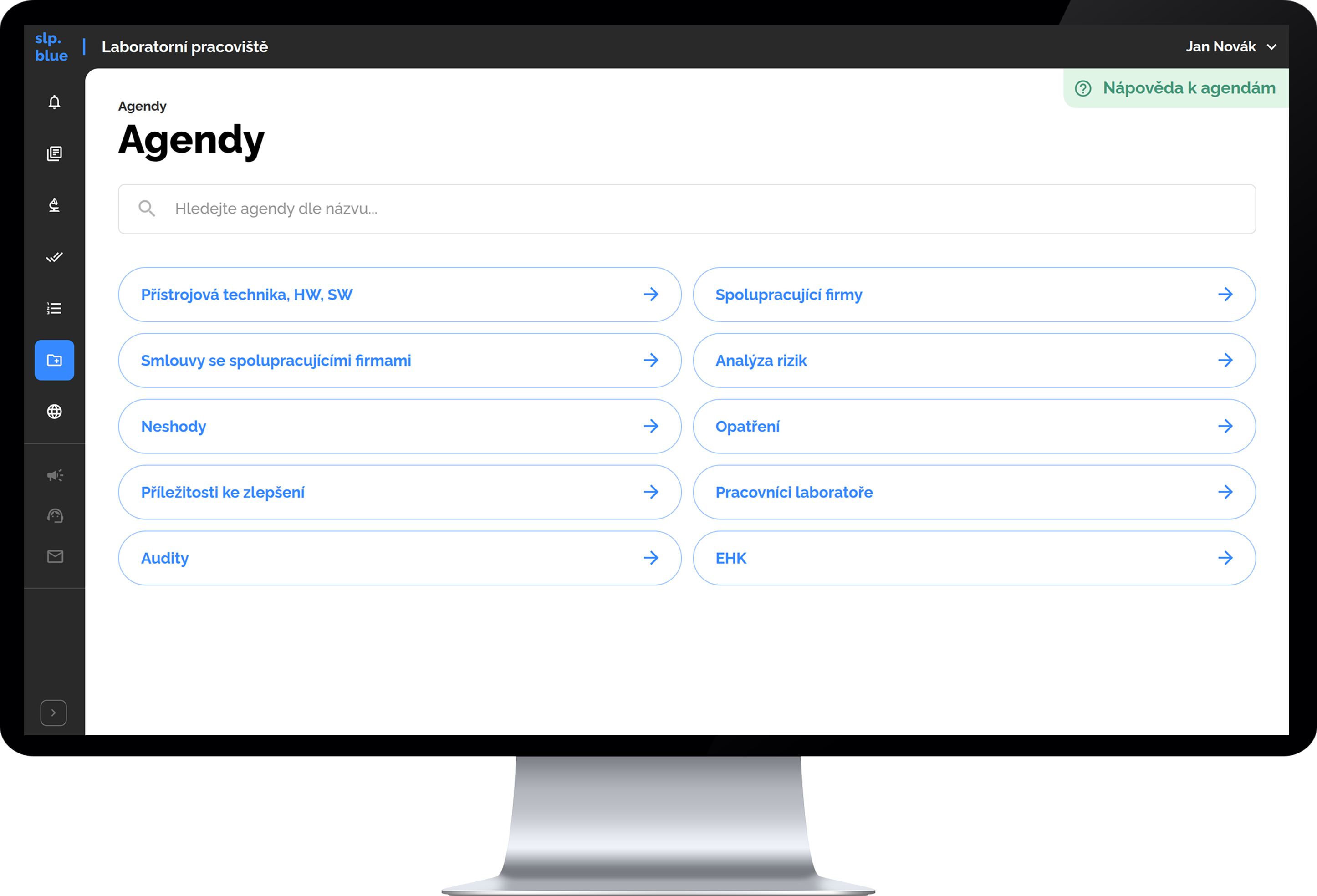
Comprehensive Quality Management Modules
- Personnel and training records
- Laboratory equipment and software
- Suppliers and their related agreements
- Risk assessments
- Nonconformities, potential improvements
- Audit planning and recording
Create Your Laboratory Web Presentation Instantly
Fast and Painless Onboarding
Whether you manage your documents using Word or another system, we migrate everything, including metadata. AI helps us to extract the data from any document format and system. We’ll even help you clean up and organize your existing documentation during the migration. Automated processes in slp.blue then keep your documentation clean and consistent — forever.
Access from Anywhere
Friendly Licensing and Ongoing Support
- Unlimited users, unlimited documents — no hidden fees or restrictions
- Automatic updates every 3 weeks with minimal downtime
- Live webinars and user feedback driving slp.blue forward
- Responsive support — we resolve issues fast because we know speed matters
Cloud or On-Premise? You Choose.
- Connect to your Laboratory Information System (LIS)
- Enable single sign-on with your existing user management system
References
Our Customers
slp.blue is currently used by medical laboratories at more than 90 sites across 50 hospitals and healthcare facilities in the Czech Republic and Slovakia. Our customers include 6 university hospitals and 22 regional hospitals. Beyond laboratories, the system is also being adopted by hospital quality departments and the first testing laboratories.
slp.blue is deployed across a wide range of specialties:
- 54× clinical biochemistry
- 39× hematology
- 20× medical microbiology
- 21× transfusion medicine
- 16× allergology and immunology
- 12× pathology
- 7× medical genetics
Laboratory Handbooks
Our customers like to use the dynamic laboratory manual feature because it allows them to always have up-to-date information in one place, easily share it via a unique URL, and do so without any involvement from the IT department.
IKEM

Centrální laboratoře FNKV

Ústav patologie FNKV

OKL Masarykovy nemocnice Rakovník
Laboratoř Krevního centra

OKBH Nemocnice Vrchlabí

OKB Nemocnice Pelhřimov

OKBMI Nemocnice Jihlava
OKB Levitova centra následné péče
Team

Tomáš Kouřim
CEO and co-founder of Mild Blue
slp.blue development coordinator

Jan Kubant
CTO and co-founder of Mild Blue
Project manager of slp.blue

Ing. Jakub Monhart
slp.blue Developer
Business Development Manager

Jan Skála
Responsible for DevOps, backend development, and slp.blue architecture

Iva Zajacová
COO
Invoices, contracts, training organisation, data import and administrative support for migrations

Pavel Mach
Head of Deployment, Instructor and Developer

Eliška Novotná
Tester and Instructor

Anastasia Surikova
Responsible for frontend development, design and UX of slp.blue

Zuzana Červínová
Support, Developer and Content specialist
in cooperation with

Antonín Jabor, Ph.D.
Deputy Head and Head of the Central Laboratory, Department of Laboratory Methods, IKEM Hospital, Prague
Head of the National Center for the Authorization of Clinical Laboratories

Jiří Kotrbatý
Quality manager and Head of the Central Reception of Samples, Department of Laboratory Methods, IKEM Hospital, Prague
Co-author of the SLP® system

Miroslav Zámečník
Co-author of the original SLP® system
Administrator of the National Laboratory Items List and Czech Medical Data Standard

Luděk Straka, Ph.D.
RNDr. Luděk Straka passed away on June 17, 2023, at the age of 60. We remember him as the main creator of the original SLP® system.
Want to learn more?
Contact us
Would you like to explore how slp.blue could be utilized in your laboratory? Feel free to contact us via e-mail at info@slp.blue or simply fill out the contact form below and we will get in touch with you soon!